
HEPATIC AND CARDIAC IRON OVERLOAD
QUANTIFICATION BY MRI AT 1.5T
O. Ernst, G. Sergent, B. Laurens, V. Gaxotte CHRU de Lille (Lille II University)
C. Rose Université Catholique de Lille (Catholic University of Lille)
Magnetic resonance imaging is based on the interaction of hydrogen nuclei with a magnetic field. An increase in the iron concentration of an organ changes the local magnetism, which, in turn, modifies the tissue signal. During imaging, the tissue signal therefore decreases when the iron concentration increases. The iron concentration can easily be quantified by measuring the decrease in the organ signal, either compared to another reference organ without any overload, such as the paravertebral muscles, or to the organ itself. This site describes the method used for the liver and the heart, particularly for thalassemias, which present with substantial overloads. The overall examination time (liver and heart) is less than 30 minutes.
If you have one of the latest Philips 1.5 T MRI devices, you can download the exam card, which lists all of the parameters used for each sequence. Please note – this is a text file. You therefore have to click the right mouse button and "save the target under".
I Quantification of hepatic iron concentration at 1.5 T
The technique is based on measuring the signal in the liver and paravertebral muscles. Multiple sequences are required to ensure optimum adaptation to the various overloads. The signal in the liver and paravertebral muscles must be recorded for each sequence. Then simply enter the values in a software program that merely applies the formula that has been determined in controls having undergone a biopsy with recording of the iron concentration. Typical formulae include a(LIVER/MUSCLE) + b, but these are valid only for a LIVER/MUSCLE ratio ranging from 0.3 to 1. Below 0.3, the background noise becomes too intense for a reliable measurement. Above 1, the signal in the liver is normal. The low range in which the LIVER/MUSCLE ratio can be used explains why several sequences with different TEs (echo times) have to be carried out.
The technique using this LIVER/MUSCLE comparison, which, in our opinion, is the most suitable, is the method described by Yves Gandon et al. It is based on 5 sequences, the TE of which varies from 21 ms to 4 ms. However, this technique cannot quantify overloads ten times greater than the normal value as the liver already has an extremely weak signal at 4 ms. We therefore supplemented this method with a sixth sequence using an extremely short TE.
PRACTICAL IMPLEMENTATION
1 Sequences
The acquisitions must be obtained using a "body coil" to ensure homogeneity. There is no need whatsoever to use a syn-body, torso or other type of surface coil. The principal parameter that must be complied with is the TE. However, compliance with the angle and the TR (response time) is also essential. Here are the 6 sequences, ranging from the most sensitive to the least sensitive, to quantify iron overload. All of these sequences have a “base” gradient echo.
SEQUENCES: parameters affecting the liver signal
1 TR: 120 ms; TE 21 ms; angle 20°
2 TR: 120 ms; TE 14 ms; angle 20°
3 TR: 120 ms; TE 9 ms; angle 20°
4 TR: 120 ms; TE 4 ms; angle 20°
5 TR: 120 ms; TE 4 ms; angle 90°
6 TR: 48 ms; TE 1.8 ms; angle 60°
SEQUENCES: The following parameters do not affect the liver signal. These parameters can, therefore, be modified.
Apnea.
Number
of sections: 8
This number cannot be used in just one acquisition with
sequence 1 without modifying the TR.
Two or three separate acquisitions
therefore have to be carried out to retain an acquisition time
compatible with apnea. Eight
sections give an overall view of the liver in order to examine a
typical, artefact-free zone. They
are spaced out:
Rectangular
matrix: 70 %.
Rectangular
visual field: 66 %.
Thickness:
10 or 15 mm.
Thick sections enhance the signal to noise ratio.
FOV 440
mm.
SEQUENCES:
technical variants to reduce
acquisition time
Multiple echoes:
the first 4 sequences differ only in
terms of TE. Two
dual-echo sequences can, therefore, be carried out instead of 4
single-echo sequences.
1
TR: 120
ms; TE 14 - 21 ms; angle 20°
2 TR:
120 ms; TE 4 - 9 ms; angle
20°
Half-Fourrier scan.
The half scan technique reduces
acquisition time by
2.
2
Measurements
The signal in the liver and muscles has to be measured in regions of interest for each sequence. Two homogeneous, artefact-free zones must be selected on the same section. Several measurements can be recorded during the same sequence to reduce the variation in results.

3 Calculating the concentration
The simplest method is to enter the values in software programs giving the iron concentration. Simply go to the following Internet page for the 5 sequences established by Yves Gandon: Hémochromatose Université de Rennes. The software will function even if you enter only one liver and muscle signal value for each sequence. If the result exceeds 330 µmol/g, you must use the sixth sequence.
This sixth sequence was created in Lille for major overloads.
The formula is as follows (µmol/g):
CHF = 937 - (537 * (liver signal / muscle signal)).
You can use the following software (MS Windows freeware) to assess the hepatic iron concentration:
4 References
Gandon Y, Guyader D, Heautot JF, Reda MI, Yaouanq J, Buhe T, Brissot P, Carsin M, Deugnier Y. Hemochromatosis: diagnosis and quantification of liver iron with gradient-echo MR imaging.Radiology. 1994 Nov;193(2):533-8.
Gandon Y, Olivie D, Guyader D, Aube C, Oberti F, Sebille V, Deugnier Y. Non-invasive assessment of hepatic iron stores by MRI.Lancet. 2004 Jan 31;363(9406):357-62.
Ernst O, Rose C, Sergent G, L'Hermine C. Hepatic iron overload: quantification with MR imaging at 1.5 T.AJR Am J Roentgenol. 1999 Apr;172(4):1141-2.
Ernst O, Sergent G, Bonvarlet P, Canva-Delcambre V, Paris JC, L'Hermine C. Hepatic iron overload: diagnosis and quantification with MR imaging.AJR Am J Roentgenol. 1997 May;168(5):1205-8
II Quantification of cardiac iron overload at 1.5 T
Generally, cardiac iron overload occurs only in the case of major overloads, particularly in the case of thalassemias. However, there is no precise correlation between biological anomalies or hepatic iron overload. Quantification of cardiac iron overload by measuring T2* has been shown to facilitate early diagnosis (1) when this T2* is below 20 ms. This measurement is also more sensitive than the measurement of ventricular function in MRI (2).
T2* is relatively straightforward to measure on a standard 1.5 T MRI device.
If a multigradient echo sequence with cardiac treegering is available (GE), the simple way is to use it.
If a multigradient echo sequence is not available (Philips), 3 gradient dual echo sequences must be carried out. They comprise two echoes so as to facilitate measurement of T2*, and are acquired with cardiac synchronization. Multiple acquisitions are required as the signal to noise ratio is low. Apnea is not, therefore, feasible. Some examinations are occasionally scattered with artefacts and are thus difficult to interpret. A “cardiac” coil is essential: A “shim” centered on the heart is essential.
1 Sequences
Cardiac
coil / Cardiac
synchronization
Number
of sections:
Just 1 section on the minor cardiac axis.
Thickness:
10 mm
FOV:
350 x 350
Matrix:
256 x 256
SENSE:
2
Sequence
1: TR/ TE-TE/
Angle: 23
ms / 2.5 - 20 ms / 20° Number of excitations per
cardiac cycle: 16
Sequence
2: TR/ TE-TE/
Angle: 13
ms / 2.5 - 10 ms / 20° Number of excitations per
cardiac cycle: 35
Sequence
3: TR/ TE-TE/
Angle: 8
ms / 2.5 - 5 ms / 20° Number of excitations per
cardiac cycle: 52
2 Measurements
The myocardial signal for each acquired echo must be measured with an ROI. The choice of ROI position is critical. The positioning must be identical for each echo of the same sequence, in an artefact-free zone. The following diagram clearly illustrates the method:

Overall, 6 signal values must, therefore, be recorded.
3 Estimation of cardiac iron concentration
In current practice, a cardiac biopsy cannot be routinely carried out to measure the iron concentration. It has not, therefore, been possible to correlate the decrease in the cardiac signal measured by T2* shortening with cardiac iron concentration. Only a T2* value can be given. If this value is below 20 ms, the cardiac signal is abnormally low, thus indicating an iron overload. The overload will obviously be higher for a T2* at 10 ms than for a T2* at 20 ms, but there is no confirmation that the concentration varies between 1 and 2 in this case. This T2* value can, however, be used to monitor the efficacy of a chelating treatment.
Calculation of T2* from the signal of 2 signal values recorded on 2 different echoes can be carried out using a scientific computer (Spreadsheet) noting that T2* corresponds to the signal decrease time constant, which follows a negative, exponential slope. Here is the formula:
T2* = (TE2 - TE1) / Log (S1 / S2) Comment: Neperian logarithm S1 First echo signal intensity T2 Second echo signal intensity
4 References
1 Anderson LJ, Holden S, Davis B, Prescott E, Charrier CC, Bunce NH, Firmin DN, Wonke B, Porter J, Walker JM, Pennell DJ. Cardiovascular T2-star (T2*) magnetic resonance for the early diagnosis of myocardial iron overload. Eur Heart J. 2001 Dec;22(23):2171-9.
2 Westwood MA, Wonke B, Maceira AM, Prescott E, Walker JM, Porter JB, Pennell DJ. Left ventricular diastolic function compared with T2* cardiovascular magnetic resonance for early detection of myocardial iron overload in thalassemia major.J Magn Reson Imaging. 2005 Aug;22(2):229-33.
III Theoretical Bases
Magnetic resonance imaging is based on the interaction between the nucleus of hydrogen atoms (single proton) and a Bo magnetic field. The technique involves positioning the patient in a strong magnetic field. To take a scan, a second magnetic B1 field is then briefly applied, the axis of which is perpendicular to that of the main Bo field. On completion of the B1 impulse, the protons take a certain length of time to regain their steady state. The very low signal transmitted by the protons during their return to steady state can only be recorded on an axis perpendicular to Bo. In fact, Bo is extremely intense, which prevents a very low signal from being recorded on this axis. The signal is, therefore, recorded in the B1 axis. A peak is reached just after completion of B1, thereafter gradually decreasing to cancel itself out within a few hundred ms. Signal cancellation follows an exponential law. We, therefore, refer to the T2 or T2* time constant, depending on the measuring technique used. The T1 time constant corresponds to the phenomenon in the main Bo magnetic axis (repelling the Bo axis) and is not involved in this exposure as the signal is captured solely on the axis perpendicular to Bo. For technical reasons, the signal is recorded only very briefly. TE, the echo time, corresponds to the interval between completion of the B1 impulse and the time of recording. The shorter the TE, the more intense is the recorded signal.

Local magnetization is increased when an organ has an elevated iron concentration. The relaxation phenomenon accelerates and the signal therefore decreases more rapidly. At an equal TE, the higher the iron concentration in an organ, the weaker the signal. To quantify the iron concentration, simply measure the signal in comparison to an organ where there is no overload, such as the paravertebral muscles, or measure the drop in signal between 2 echoes to calculate the T2 or T2*. The higher the iron concentration, the lower the T2 or T2*. Some investigators use R2 = 1/T2 or R2* = 1/T2* values to obtain a variation parallel to the iron concentration. This is a different, albeit straightforward, way of presenting the results. The following diagram clearly illustrates these mechanisms.
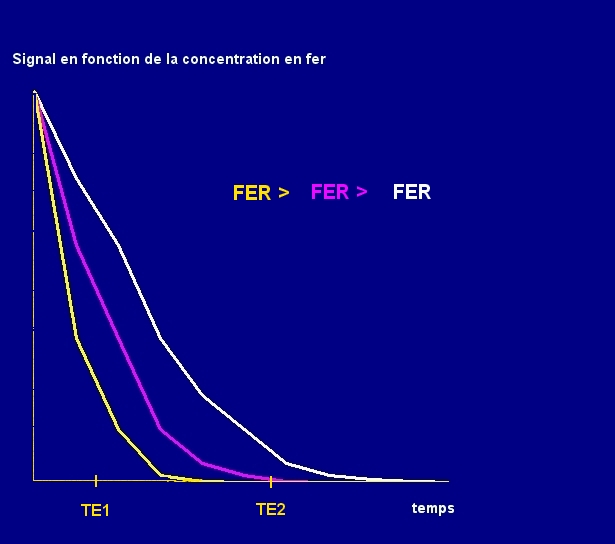
fer = iron
This diagram shows that, the higher its iron concentration, the faster the decrease in the organ signal after the B1 impulse. For the first echo time (TE1), the signal is not zero, regardless of the concentration. Nevertheless, it is already weak for a high concentration (yellow). For the second echo time (TE2), there is no longer any signal for the 2 cases of iron overload (yellow and violet). Only the normal organ (white) continues to transmit a signal.
Two methods can be used to quantify iron. In the first method, the organ signal is compared to a reference organ that does not present any overload, such as the paravertebral muscles. This comparison requires the organ to have a signal that is already weak, but which can still be measured easily, i.e. greater than the background noise. Ideally, it must range between 0.3 and 0.8 times the normal organ signal. Multiple acquisitions with TEs of different values must, therefore, be carried out in order to ensure optimal adaptation to the various potential iron concentrations. The second technique involves measuring the signal ratio between 2 echoes to calculate the T2*. Once again, multiple echoes are essential in order to adapt to the iron concentration. Regardless of the method used, the results obtained must be compared to those of an experimental series in which the concentration was measured on biopsy in order to plot a calibration curve. This type of curve exists for the liver, but not for the heart. In the liver, it is, therefore, possible to establish a genuine iron concentration whereas, in the heart, only a T2* can be given.
O. Ernst, G. Sergent, B. Laurens, V. Gaxotte CHRU de Lille (Lille II University)
C. Rose Université Catholique de Lille (Catholic University of Lille)
December 2005
Key words: hemochromatosis / MRI / nuclear magnetic resonance imaging / hepatic siderosis / hemosiderosis / cardiac siderosis / iron / thalassemia